
Cyanation of Aryl Halides
The preparation of aryl cyanides is one of those reactions that med chemists do sooner rather than later. Many protocols are available, from the old diazocompounds Sandmeyer reaction to modern Cu and Pd mediated methods. However, though the Copper mediated methods are well developed there is still a need of improved Palladium cyanation methods.
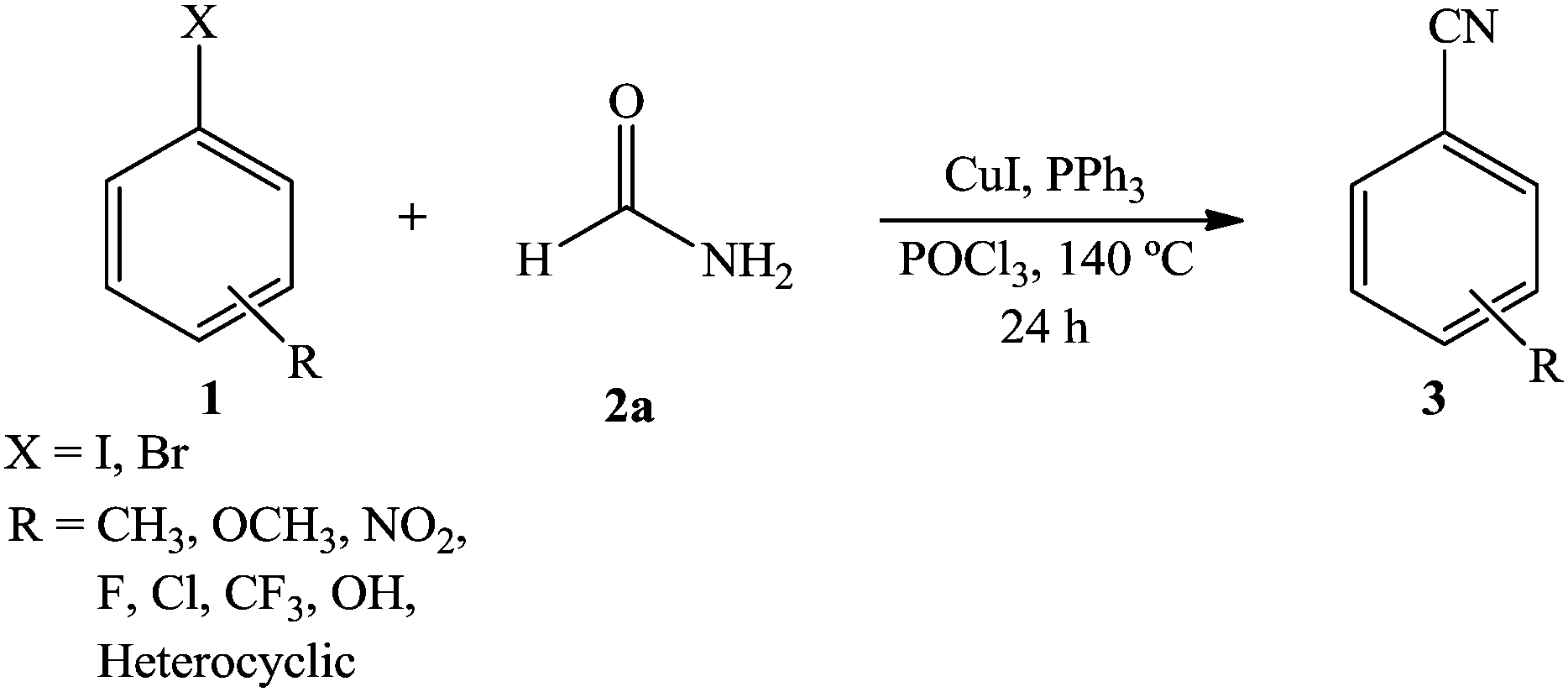
RSC Adv., 2014,4, 13405-13408
DOI: 10.1039/C3RA48075E
A copper iodide/triphenyl phosphine catalyzed simple and efficient protocol has been developed for cyanide free cyanation of aryl halide using formamide as a cyanide source. The reaction works well to furnish aryl nitriles using an inexpensive and easily available copper catalyst with triphenyl phosphine and phosphorus oxychloride. Under the optimized reaction conditions, a variety of electron-donating and electron-withdrawing aryl halides were efficiently converted into the respective nitriles in moderate to excellent yields.
http://newdrugapprovals.org/
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTOBelow paper by Lilly chemists the chosen Pd source is Pd on carbon, which is unusual, and the cyanide source is Zn(CN)2. The protocol involves heating a mixture of the aryl halide (typically a bromide, though chlorides can be used also) with Zn(CN)2 (60 mol%), Pd/C (2 mol%), dppf (4 mol%) and Zn(CHO2)2·H2O (10 mol%) in DMAC. The mixture is heated to 100-120 °C for 12 h. For conditions that look really harsh, yields are surprisingly good, ranging from 77 to 96%. Many examples with heterocycles are included.
Development of Pd/C-Catalyzed Cyanation of Aryl Halides J. Org. Chem., 2011, 76 (2), pp 665–668. See:10.1021/jo102037y
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTOSee below paper by Kwong et al. (The Hong Kong Polytechnic University, China) follows similar guidelines, but with a very different outcome. Their protocol involves heating a mixture of the aryl halide, specifically aryl chlorides with K4[Fe(CN)6]·3H2O, Pd(OAc)2 (2 mol%), CM-phos (4 mol%) and Na2CO3 (12 mol%) in acetonitrile/water at 70 °C for 18 h. They report 16 examples, including several heterocycles, with yields ranging from 74 to 97%.
I must add that it is a very nice paper. The authors have carried out a screening of ligands, they have studied the effect of base and solvent on the outcome of the reaction and they include even some data on how the water content of the solvent mixture affects the yield, all very useful if someone wants to scale up this reaction. In this case, an added bonus of this protocol is the low toxicity of K4[Fe(CN)6]·3H2O compared to the highly toxic Zn(CN)2.
A Mild and Efficient Palladium-Catalyzed Cyanation of Aryl Chlorides with K4[Fe(CN)6] Org. Lett., 2011, 13(4), pp 648–651. See: 10.1021/ol1028892
http://newdrugapprovals.org/
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTOPalladium-catalyzed Cyanation of Aryl Halides
Cheng et al. have reported the palladium-catalyzed cyanation of aryl halides. According to their results, in the presence of [1,2-bis(diphenylphosphino)ethane]palladium(II) dichloride catalyst, formic acid and sodium formate as an acid and an additive, respectively, aryl halides react with cuprous thiocyanate to provide the corresponding aromatic nitriles in good yields. In this reaction, even when using aryl boronic acids instead of aryl halides, the reaction successfully proceeds and the dihydroxyboryl groups are directly replaced with the cyano group. Since cuprous thiocyanate is an inexpensive and nontoxic cyanide source, this protocol is a novel and safe method to access aromatic nitriles.

http://newdrugapprovals.org/
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
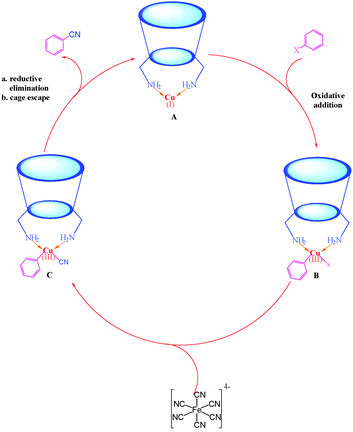
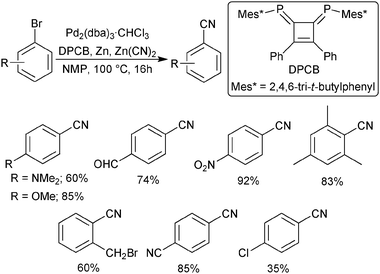
http://pubs.rsc.org/en/content/articlelanding/2011/cs/c1cs15004a#!divAbstract

http://onlinelibrary.wiley.com/doi/10.1002/anie.201304188/abstract
http://www.arkat-usa.org/arkivoc-journal/browse-arkivoc/2010/6/graphical-abstracts/
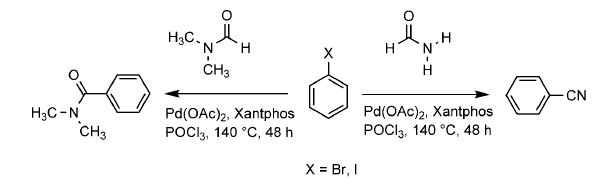
http://www.chemistryviews.org/details/news/1033591/Cyanide-Free_Cyanation.html

http://organicsynthesisinternational.blogspot.in/2013/05/copper-mediated-cyanation-of-aryl.html
Read all about Organic Spectroscopy on ORGANIC SPECTROSCOPY INTERNATIONAL 

http://newdrugapprovals.org/
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO


No comments:
Post a Comment