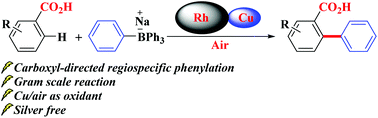
Rhodium-catalyzed regiospecific C-H ortho-phenylation of benzoic acids with Cu/air as an oxidant
Rhodium-catalyzed regiospecific C–H ortho-phenylation of benzoic acids with Cu/air as an oxidant
*
Corresponding authors
a
The Key Laboratory of Environmentally Friendly Chemistry and Application of the Ministry of Education, College of Chemistry, Xiangtan University, Xiangtan 411105, China
E-mail: hgong@xtu.edu.cn
E-mail: hgong@xtu.edu.cn
b
Department of Chemistry and FQRNT Centre for Green Chemistry and Catalysis, McGill University, 801 Sherbrooke St. W., Montreal, Canada
E-mail: cj.li@mcgill.ca
E-mail: cj.li@mcgill.ca
Org. Chem. Front., 2017, Advance Article
DOI: 10.1039/C6QO00663A
http://pubs.rsc.org/en/Content/ArticleLanding/2017/QO/C6QO00663A#!divAbstract
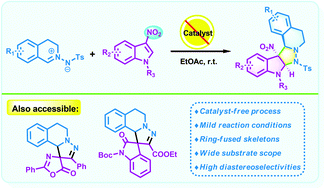
The development of an efficient and practical synthetic approach for the regiospecific C–H ortho-phenylation of aromatic carboxylic acids without the use of silver or costly coupling partners is reported. The rhodium-catalyzed C–H phenylation proceeded in the presence of Cu/air as a terminal oxidant. In most cases, numerous inactivated benzoic acids could couple with low-cost NaBPh4 to obtain good to excellent yields.
A solution of aromatic acid 1a (27.2 mg, 0.2 mmol), NaBPh4 (0.27 g, 0.8 mmol), KF (46 mg, 0.8 mmol), [Rh(nbd)Cl]2 (9.2 mg, 0.02 mmol) and CuBr2 (4.5 mg, 0.02 mmol) in chlorobenzene (1.2 mL) was stirred in a sealed tube under air at 150 oC for 24 h. The reaction mixture was then cooled to room temperature and acidified by dilute aqueous HCl to pH<3, and then the solvent was evaporated in vacuo. The residue was purified by preparative thin-layer chromatography (TLC) on silica gel with petroleum ether and ethyl acetate as eluent to give the pure product 2a.
2a White solid; Yield 86%; Mp 132-134 oC [lit1 mp 133-135oC]; IR (neat) νmax 3057, 2917, 2849, 2627, 1682, 1461, 1133, 1064, 1000, 759, 696 cm-1; 1H NMR (400 MHz, CDCl3): δ = 7.35-7.40 (m, 6H), 7.23 (m, 2H), 2.45 (s, 3H); 13C NMR (100 MHz, CDCl3) δ = 174.5, 140.7, 140.2, 135.5, 132.2, 129.7, 129.2, 128.4 (2C), 127.6, 127.5, 20.0; HRMS (ESI) m/z calcd for C14H13O2 213.0910, found [M+H] + 213.0912.
//////////








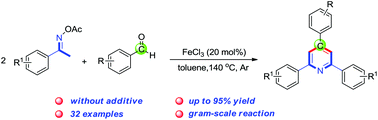



 New class of cyclic Weinreb amides for oxidative C-H
olefination or Heck reaction
New class of cyclic Weinreb amides for oxidative C-H
olefination or Heck reaction