THIS PAGE IS PART 2
PART 1...........http://drugsynthesisint.blogspot.in/p/gliptin-series.html
1.TRELAGLIPTIN
2.OMARIGLIPTIN
3.RETAGLIPTIN
4 CARMEGLIPTIN
5 IMIGLIPTIN
6 (S) SITAGLIPTIN
7 DENAGLIPTIN
8 DUTOGLIPTIN
SEE
9 TENELIGLIPTIN
10 GOSOGLIPTIN
11 EVOGLIPTIN
12 MELOGLIPTIN
13 SITAGLIPTIN
14 SAXAGLIPTIN
15
16
17
PART 2 AT.......http://organicsynthesisinternational.blogspot.in/p/gliptin-series-22.html
will be updated
P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent.

9
Teneligliptin
http://www.allfordrugs.com/2015/07/03/teneligliptin/
OR
http://newdrugapprovals.org/2015/07/03/teneligliptin/

10 Gosogliptin
http://www.allfordrugs.com/2015/07/03/gosogliptin/
OR
http://newdrugapprovals.org/2015/07/03/gosogliptin/

11
EVOGLIPTIN
EVOGLIPTIN
CAS: 1222102-29-5 FREE
HCL……
Dong-A Pharmaceutical. Co., Ltd, 동아제약 주식회사
2-Piperazinone, 4-((3R)-3-amino-1-oxo-4-(2,4,5-trifluorophenyl)butyl)-3-((1,1-dimethylethoxy)methyl)-, (3R)-
R)-4-((R)-3-Amino-4-(2,4,5-trifluorophenyl)-butanoyl)-3-(t-butoxymethyl)-piperazin-2-one
4-[3(R)-Amino-4-(2,4,5-trifluorophenyl)butyryl]-3(R)-(tert-butoxymethyl)piperazin-2-one hydrochloride
DA-1229
DA-1229 is a dipeptidyl peptidase IV (CD26) inhibitor currently being developed in phase III clinical studies at Dong-A for the treatment of type 2 diabetes.
In 2014, Eurofarma aquired rights for product development and commercialization in Brazil.

86…………H. J. Kim, W. Y. Kwak, J. P. Min, J. Y. Lee, T. H. Yoon, H. D. Kim, C. Y. Shin, M. K.
Kim, S. H. Choi, H. S. Kim, E. K. Yang, Y. H. Cheong, Y. N. Chae, K. J. Park, J. M.
Jang, S. J. Choi, M. H. Son, S. H. Kim, M. Yoo and B. J. Lee, Bioorg. Med. Chem. Lett.,
2011, 21 (12), 3809-3812.
[87] …………K. S. Lim, J. Y. Cho, B. H. Kim, J. R. Kim, H. S. Kim, D. K. Kim, S. H. Kim, H. J. Yim,
S. H. Lee, S. G. Shin, I. J. Jang and K. S. Yu, Br. J. Clin. Pharmacol., 2009, 68 (6), 883-
890.
- Originator Dong-A Pharmaceutical
- Developer Dong-A ST
- Class Amides; Antihyperglycaemics; Fluorobenzenes; Piperazines; Small molecules
- Mechanism of Action CD26 antigen inhibitors
- Orphan Drug Status No
- On Fast track No
- New Molecular Entity Yes
- Available For Licensing Yes – Type 2 diabetes mellitus
Highest Development Phases
- Phase III Type 2 diabetes mellitus
Most Recent Events
- 01 Sep 2014 Phase-I clinical trials in Type-2 diabetes mellitus (In volunteers) in United Kingdom (PO)
- 31 Jul 2014 Phase-III clinical trials in Type-2 diabetes mellitus in South Korea (PO)
- 31 Jul 2014 Dong-A ST initiates enrolment in a phase I trial in patients with renal impairment in South Korea (NCT02214693)

…………………………………..
WO 2010114291
http://www.google.co.in/patents/WO2010114291A2?cl=en
Formula 1
Korea Patent Publication No. 2008-0094604 the call to the scheme, as indicated by A Ⅰ) of formula (II) beta-compound of formula 3 is already substituted heterocyclic compound having 1-hydroxy-benzotriazole group (HOBT) 1-ethyl-3- (3-dimethylaminopropyl) carbodiimide (EDC) and reacting with a tertiary amine to prepare a compound of formula (4) connected by peptide bonds; Ⅱ) beta comprises the step of reacting under acidic conditions a compound of the formula (4) – a method of manufacturing the heterocyclic compounds of the formula I having an amino group is disclosed.
- Scheme A]
(Wherein, PG is a protecting group.)
In this case, the beta of the formula (2) of Scheme A – a compound having an amino group is prepared in addition to the DPP-IV inhibitor International Publication represented by Formula 1 WO03 / 000181, WO03 / 004498, WO03 / 082817, WO04 / 007468, WO04 / 032836, WO05 / 011581, WO06 / 097175, WO07 / 077508, WO07 / 063928, WO08 / 028662 WO08 / it may be used for the production of different DPP-IV inhibitors according 087,560 and can be prepared in a number of ways.
To, the compound of Formula 2 is an example as shown in Scheme J. Med.Chem. 2005; 141, and Synthesis 1997; it can be produced by the known method described in 873.
Specifically, (2S) – (+) – 2,5- dihydro-3,6-dimethoxy-2-isopropyl-pyrazine 2,4,5-trifluoro-react with benzyl bromide and acid treatment, and then the amine an ester compound obtained by the protection reaction. Ester compounds are hydrolyzed to re-3- (2,4,5-trifluoro-phenyl) -2-amino-propionic acid tert such as isobutyl chloroformate, triethylamine or diisopropylethylamine to give the amine, and then using diazomethane to form a diazo ketone, and then may be prepared by reaction with silver benzoate. However, the reaction can be performed at low temperature (-78 ℃) or high alpha-amino acid to purchase and use, and may have a risk of problems such as the need to use large diazomethane.
To a different process for preparing a compound of Formula 2 as shown in scheme Tetrahedron: Asymmetry 2006; It is known in 2622; 205 or similarly Bioorganic & Medicinal Chemistry Letters 2007.
That is, a 1,1′-carbonyl-2,4,5 which the phenyl trifluoroacetic acid activated using the following imidazole mono-methyl words potassium carbonate is reacted with the beta-keto ester compound is prepared. This produced an enamine ester using ammonium acetate and ammonium solution, the ester compound chloro (1,5-cyclooctadiene) rhodium (I) dimer using a chiral ferrocenyl ligands I the reaction of the high-pressure hydrogen with a chiral primary amine with a beta-amino ester compound after production and can lead to hydrolysis to prepare a compound of formula (2). However, use of expensive metal catalyst has a problem that must be performed in high pressure hydrogenation.
The method for preparing a compound of Formula 2 is disclosed in International Publication No. WO 04/87650.
Specifically, 2,4,5-fluorophenyl reagent is oxalyl chloride, the acid activated acid with 2,2-dimethyl-1,3-dioxane-4,6-dione, and after the reaction of methanol and the resulting material at reflux to prepare a corresponding compound. With a selective reducing reagents which enantiomers (S) -BINAP-RuCl 2 and hydrogen through a reaction (S) – producing a compound having coordinated to each other, it again after the decomposition, and the singer O- benzyl hydroxyl amine and the coupling reaction and the intermediate is prepared. To do this, the resulting intermediate tree azodicarboxylate and diisopropyl azodicarboxylate presence ring condensation reaction, treated with an aqueous solution of lithium hydroxide to (R) – while having the formula (II) coordinated to the amine group protected with a benzyl-O- the compound can be produced. However, the method has a problem as a whole to be prepared by the reaction yield to be low and a long processing time to perform the reaction.
Thus, the conventional known method for producing a compound of the general formula (2) has the disadvantage of using expensive reagents, or not suitable for commercial mass-production method by a long synthesis time yield is also low.
In addition, the compound represented by General Formula (3), as described in Korea Patent Publication No. 2008-0094604 call, can be prepared by way of reaction schemes.
Specifically, the starting material D- serine methyl ester is substituted by a hydroxy group when reflux again substituted by trityl chloride as methoxy groups converted to the aziridine compound.
[Scheme 3]
<Example 3> (R)-4-[(R)-3-아미노-4-(2,4,5-트리플루오로페닐)부타노일]-3-(t-부톡시메틸)피페라진-2-온(화학식 1) Preparation of the hydrochloride
Step 1: t- butyl (R)-4-[(R)-2-(t-부톡시메틸)-3-옥소피페라진-1-일]-4-옥소 - 1-(2,4,5-트리플루오로페닐)부탄-2-일카르바메이트(화학식 Preparation of 4)
2 L flask, prepared in Example 1 (R) -3-t- butoxycarbonyl-4- (2,4,5-trifluoro-phenyl) butanoate acid (Formula 2) 10.0 g of toluene was dissolved in 450 mL of bis (2,2′-benzothiazolyl) disulfide 13.0 g, was cooled and then 10.2 g triphenylphosphine was added to the reaction solution at 0 ℃. While stirring the reaction mixture was added to a solution of 0.8 mL of triethylamine in 20 mL of toluene was stirred at room temperature for 5 hours. The reaction mixture was cooled to 0 ℃ and prepared in Example 2 (R) -3- (t- butoxymethyl) piperazin-2-one (Formula 3) was dissolved in 5.6 g of toluene and 40 mL pyridine a 2.4 mL was added slowly. After 30 minutes the reaction mixture was heated to room temperature and stirred for 1 hour. Saturated sheet to be the aqueous acid solution to a pH of 2.5 and then diluted with ethyl acetate 400 mL. Washed twice with brine and the organic layer was dehydrated with magnesium sulfate and concentrated. The residue was purified by column chromatography to give the title compound 838 mg.
1 H NMR (400 MHz, CDCl 3) δ 7.03 (m, 1H), 6.88 (m, 1H), 5.97 (m, 1H), 5.48 (m, 1H), 4.16 ~ 4.07 (m, 1H), 4.02 ~ 3.91 (m, 1H), 3.74 (m, 2H) 3.37 (m, 2H), 3.24 (m, 1H), 2.92 (m, 2H), 2.80 (m, 1H), 2.59 (m, 2H), 1.34 ( d, 9H), 1.13 (s, 9H)
Step 2: (R) -4 – [(R) -3- amino-4- (2,4,5-trifluoro-phenyl) butane five days] -3- (t- butoxymethyl) piperazin-2- on the production of (I) hydrochloride
Prepared in Step 1 t- butyl (R)-4-[(R)-2-(t-부톡시메틸)-3-옥소피페라진-1-일]-4-옥소-1-(2,4,5-트리플루오로페닐)부탄-2-일 카르바메이트 97 mg was dissolved in methanol was added 3 mL 2N- hydrochloric acid / diethyl ether 2 mL was stirred at room temperature for 3 hours. The reaction mixture was concentrated and dried under reduced pressure to give 64 mg of the title compound as a foaming solid.
1 H NMR (400 MHz, CD 3 OD) δ 7.37 (m, 1H), 7.23 (m, 1H), 4.80 (m, 1H), 4.59 ~ 4.40 (m, 1H), 3.93 (m, 1H), 3.90 ~ 3.83 (m, 2H), 3.70 (m, 1H), 3.38 (m, 2H), 3.27 (m, 1H), 3.07 (m, 2H), 2.89 ~ 2.66 (m, 2H), 1.18 (s, 3H ), 1.11 (s, 6H)
Mass (M + 1): 402
<Example 4> (R)-4-[(R)-3-아미노-4-(2,4,5-트리플루오로페닐)부타노일]-3-(t-부톡시메틸)피페라진-2-온(화학식 1) tartaric acid salts
Step 1: (R) -4 – [(R) -3- amino-4- (2,4,5-trifluoro-phenyl) butane five days] -3- (t- butoxymethyl) piperazin-2- Preparation of one (I)
Example 3 to give a compound of formula I in hydrochloride 60 mg 5% sodium hydrogen carbonate in dichloromethane was added to 10 mL of an aqueous solution / 2-propanol (4/1 (v / v)) was added to the mixed solution and extracted two times 10 mL The organic layer was dried under reduced pressure to give 55 mg of the title compound as a solid.
1 H NMR (400 MHz, CD 3 OD) δ 7.27 (m, 1H), 7.14 (m, 1H), 4.56 ~ 4.39 (m, 1H), 3.96 ~ 3.81 (m, 3H), 3.70 (m, 1H) , 3.46 (m, 1H), 3.43 ~ 3.32 (m, 1H), 2.83 ~ 2.65 (m, 3H), 2.58 ~ 2.40 (m, 2H), 1.16 (s, 3H), 1.11 (s, 6H)
Mass (M + 1): 402
Step 2: (R) -4 – [(R) -3- amino-4- (2,4,5-trifluorophenyl) butanoyl] -3- (t- butoxymethyl) piperazin-2- one (I) tartaric acid salt [
Was dissolved 55 mg of the compound of step 1 in 0.56 mL of acetone, L- tartrate 26 mg ethanol / water (9/1 (v / v)) was added slowly to a solution of 0.35 mL was stirred for 30 minutes. Here was added 0.56 mL of 2-propanol was stirred for 10 minutes and re-filtered to give 77 mg of the title compound as a solid.
1 H NMR (400 MHz, CD 3 OD) δ 7.38 (m, 1H), 7.22 (m, 1H), 4.80 (m, 1H), 4.59 ~ 4.40 (m, 1H), 4.40 (s, 2H), 3.93 (m, 1H), 3.90 ~ 3.83 (m, 2H), 3.70 (m, 1H), 3.38 (m, 2H), 3.27 (m, 1H), 3.07 (m, 2H), 2.89 ~ 2.66 (m, 2H ), 1.15 (s, 3H), 1.11 (s, 6H)
Mass (M + 1): 402
………………………………Step 1: t- butyl (R)-4-[(R)-2-(t-부톡시메틸)-3-옥소피페라진-1-일]-4-옥소 - 1-(2,4,5-트리플루오로페닐)부탄-2-일카르바메이트(화학식 Preparation of 4)
2 L flask, prepared in Example 1 (R) -3-t- butoxycarbonyl-4- (2,4,5-trifluoro-phenyl) butanoate acid (Formula 2) 10.0 g of toluene was dissolved in 450 mL of bis (2,2′-benzothiazolyl) disulfide 13.0 g, was cooled and then 10.2 g triphenylphosphine was added to the reaction solution at 0 ℃. While stirring the reaction mixture was added to a solution of 0.8 mL of triethylamine in 20 mL of toluene was stirred at room temperature for 5 hours. The reaction mixture was cooled to 0 ℃ and prepared in Example 2 (R) -3- (t- butoxymethyl) piperazin-2-one (Formula 3) was dissolved in 5.6 g of toluene and 40 mL pyridine a 2.4 mL was added slowly. After 30 minutes the reaction mixture was heated to room temperature and stirred for 1 hour. Saturated sheet to be the aqueous acid solution to a pH of 2.5 and then diluted with ethyl acetate 400 mL. Washed twice with brine and the organic layer was dehydrated with magnesium sulfate and concentrated. The residue was purified by column chromatography to give the title compound 838 mg.
1 H NMR (400 MHz, CDCl 3) δ 7.03 (m, 1H), 6.88 (m, 1H), 5.97 (m, 1H), 5.48 (m, 1H), 4.16 ~ 4.07 (m, 1H), 4.02 ~ 3.91 (m, 1H), 3.74 (m, 2H) 3.37 (m, 2H), 3.24 (m, 1H), 2.92 (m, 2H), 2.80 (m, 1H), 2.59 (m, 2H), 1.34 ( d, 9H), 1.13 (s, 9H)
Step 2: (R) -4 – [(R) -3- amino-4- (2,4,5-trifluoro-phenyl) butane five days] -3- (t- butoxymethyl) piperazin-2- on the production of (I) hydrochloride
Prepared in Step 1 t- butyl (R)-4-[(R)-2-(t-부톡시메틸)-3-옥소피페라진-1-일]-4-옥소-1-(2,4,5-트리플루오로페닐)부탄-2-일 카르바메이트 97 mg was dissolved in methanol was added 3 mL 2N- hydrochloric acid / diethyl ether 2 mL was stirred at room temperature for 3 hours. The reaction mixture was concentrated and dried under reduced pressure to give 64 mg of the title compound as a foaming solid.
1 H NMR (400 MHz, CD 3 OD) δ 7.37 (m, 1H), 7.23 (m, 1H), 4.80 (m, 1H), 4.59 ~ 4.40 (m, 1H), 3.93 (m, 1H), 3.90 ~ 3.83 (m, 2H), 3.70 (m, 1H), 3.38 (m, 2H), 3.27 (m, 1H), 3.07 (m, 2H), 2.89 ~ 2.66 (m, 2H), 1.18 (s, 3H ), 1.11 (s, 6H)
Mass (M + 1): 402
<Example 4> (R)-4-[(R)-3-아미노-4-(2,4,5-트리플루오로페닐)부타노일]-3-(t-부톡시메틸)피페라진-2-온(화학식 1) tartaric acid salts
Step 1: (R) -4 – [(R) -3- amino-4- (2,4,5-trifluoro-phenyl) butane five days] -3- (t- butoxymethyl) piperazin-2- Preparation of one (I)
Example 3 to give a compound of formula I in hydrochloride 60 mg 5% sodium hydrogen carbonate in dichloromethane was added to 10 mL of an aqueous solution / 2-propanol (4/1 (v / v)) was added to the mixed solution and extracted two times 10 mL The organic layer was dried under reduced pressure to give 55 mg of the title compound as a solid.
1 H NMR (400 MHz, CD 3 OD) δ 7.27 (m, 1H), 7.14 (m, 1H), 4.56 ~ 4.39 (m, 1H), 3.96 ~ 3.81 (m, 3H), 3.70 (m, 1H) , 3.46 (m, 1H), 3.43 ~ 3.32 (m, 1H), 2.83 ~ 2.65 (m, 3H), 2.58 ~ 2.40 (m, 2H), 1.16 (s, 3H), 1.11 (s, 6H)
Mass (M + 1): 402
Step 2: (R) -4 – [(R) -3- amino-4- (2,4,5-trifluorophenyl) butanoyl] -3- (t- butoxymethyl) piperazin-2- one (I) tartaric acid salt [
Was dissolved 55 mg of the compound of step 1 in 0.56 mL of acetone, L- tartrate 26 mg ethanol / water (9/1 (v / v)) was added slowly to a solution of 0.35 mL was stirred for 30 minutes. Here was added 0.56 mL of 2-propanol was stirred for 10 minutes and re-filtered to give 77 mg of the title compound as a solid.
1 H NMR (400 MHz, CD 3 OD) δ 7.38 (m, 1H), 7.22 (m, 1H), 4.80 (m, 1H), 4.59 ~ 4.40 (m, 1H), 4.40 (s, 2H), 3.93 (m, 1H), 3.90 ~ 3.83 (m, 2H), 3.70 (m, 1H), 3.38 (m, 2H), 3.27 (m, 1H), 3.07 (m, 2H), 2.89 ~ 2.66 (m, 2H ), 1.15 (s, 3H), 1.11 (s, 6H)
Mass (M + 1): 402
WO 2010114292
http://www.google.com/patents/WO2010114292A2?cl=en
…………………………………
Discovery of DA-1229: a potent, long acting dipeptidyl peptidase-4 inhibitor for the treatment of type 2 diabetes
Bioorg Med Chem Lett 2011, 21(12): 3809
http://www.sciencedirect.com/science/article/pii/S0960894X11004859

A series of β-amino amide containing substituted piperazine-2-one derivatives was synthesized and evaluated as inhibitors of dipeptidyl pepdidase-4 (DPP-4) for the treatment of type 2 diabetes. As results of intensive SAR study of the series, (R)-4-[(R)-3-amino-4-(2,4,5-trifluorophenyl)-butanoyl]-3-(t-butoxymethyl)-piperazin-2-one (DA-1229) displayed potent DPP-4 inhibition pattern in several animal models, was selected for clinical development.
About evogliptin tartrate tablets
Evogliptin tartrate tablets is a dipeptidyl peptidase IV inhibitor, in tablet form. Evogliptin tartrate
tablets is expected to be approved for the treatment of type 2 diabetes mellitus. The Group holds
an exclusive intellectual property licence from Dong-A Pharmaceutical Co. Ltd. to develop
and commercialise evogliptin tartrate tablets in China, including the exclusive right to develop
evogliptin tartrate tablets for manufacturing and sale in the Group’s name. The new drug certificate
to be issued by the CFDA will be approved and registered under the Group’s name.
Evogliptin is a patented new molecular entity in the United States and other international markets.
Evogliptin tartrate tablets is being concurrently developed by Dong-A Pharmaceutical Co. Ltd.
for the Korean market. Based on information released from a multi-centre, phase II, randomised,
double-blind, placebo-controlled, therapeutic exploratory clinical trial conducted in Korea by
Dong-A Pharmaceutical Co. Ltd. to investigate the efficacy and safety of evogliptin, evogliptin
was proven to be effective in significantly lowering blood glucose levels in patients with type
2 diabetes. Data also show that the body weights of patients remain stable over the treatment
period. In addition, evogliptin was proven to be safe and well tolerated with no severe adverse
drug reactions observed during those phase II clinical trials. The Company believes evogliptin
tartrate tablets will help reduce the burden of patients with moderate-to-severe renal impairment
as pharmacokinetic study in animal model and healthy human volunteers showed low renal
elimination.
2
According to the statistics of IMS Health Incorporated, the market size of products for the
treatment of diabetes in China in 2013 was approximately RMB7.8 billion, and grew at a
compound annual growth rate of 23.4% from 2011 to 2013.
http://www.luye.cn/en/uploads//2014-07/21/_1405936452_zr21xh.pdf
Dong-A ST
SEOUL, SOUTH KOREA
14 April 2015 – 5:45pm
Oh Seung-mock
Dong-A ST has licensed its new diabetes drug Evogliptin to 17 Latin
American countries including Mexico, Venezuela, Argentina, Chile,
Colombia, Ecuador, Peru, the Dominican Republic, and Uruguay, Jung
Jae-wook, Dong-A ST’s PR manager, told Business Korea.
Dong-A ST and Eurofarma, a Brazilian pharmaceutical company, concluded the licensing contract at Dong-A ST’s headquarters on April 13 in Seoul.
Eurofarma will be responsible for Evogliptin’s product development and sales in the 17 Latin American countries, Dong-A ST said. Dong-A ST will receive royalties from Eurofarma, and export the raw material of the medicine.
Dong-A ST has been developing Evogliptin with the support of the Ministry of Health & Welfare of South Korea as an innovative new medicine research project since May 2008. Evogliptin is a DPP-4 remedy based on the inhibition mechanism which is “excellent” at reducing blood sugar, whilst “less likely” to cause weight increases and hypoglycemia, the company said.
Park Chan-il, president of Dong-A ST, said that Dong-A ST will pursue further out-licensing “over the globe,” through continuous investment in research and development.
Maurizio Billi, Eurofarma’s president, wished to expand both companies’ partnership in the innovative new remedy development sector, according to Dong-A ST.
Last July, Dong-A ST and Eurofarma concluded a contract out-licensing Evogliptin to Brazil itself, the company said.
Dong-A ST and Eurofarma, a Brazilian pharmaceutical company, concluded the licensing contract at Dong-A ST’s headquarters on April 13 in Seoul.
Eurofarma will be responsible for Evogliptin’s product development and sales in the 17 Latin American countries, Dong-A ST said. Dong-A ST will receive royalties from Eurofarma, and export the raw material of the medicine.
Dong-A ST has been developing Evogliptin with the support of the Ministry of Health & Welfare of South Korea as an innovative new medicine research project since May 2008. Evogliptin is a DPP-4 remedy based on the inhibition mechanism which is “excellent” at reducing blood sugar, whilst “less likely” to cause weight increases and hypoglycemia, the company said.
Park Chan-il, president of Dong-A ST, said that Dong-A ST will pursue further out-licensing “over the globe,” through continuous investment in research and development.
Maurizio Billi, Eurofarma’s president, wished to expand both companies’ partnership in the innovative new remedy development sector, according to Dong-A ST.
Last July, Dong-A ST and Eurofarma concluded a contract out-licensing Evogliptin to Brazil itself, the company said.

12
MELOGLIPTIN
GRC 8200; 868771-57-7, EMD-675992
4-fluoro-1-[2-[[(1R,3S)-3-(1,2,4-triazol-1-ylmethyl)cyclopentyl]amino]acetyl]pyrrolidine-2-carbonitrile
4(S)-Fluoro-1-[2-[(1R,3S)-3-(1H-1,2,4-triazol-1-ylmethyl)cyclopentylamino]acetyl]pyrrolidine-2(S)-carbonitrile
GRC-8200, a dipeptidyl peptidase IV inhibitor (DPP-IV), is currently undergoing phase II clinical trials at Glenmark Pharmaceuticals and Merck KGaA for the treatment of type 2 diabetes. In 2006, the compound was licensed by Glenmark Pharmaceuticals to Merck KGaA in Europe, Japan and N. America for the treatment of type 2 diabetes, however, these rights were reaquired by Glenmark in 2008.
ALTERNATE……….
13

SITAGLIPTIN
http://orgspectroscopyint.blogspot.in/2015/03/sitagliptin.html
http://www.allfordrugs.com/2015/07/05/sitagliptin/
P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent.
14

SAXAGLIPTIN

SAXAGLIPTIN
| Saxagliptin | |
| CAS No.: | 361442-04-8 |
|---|---|
| Synonyms: |
|
| Formula: | C18H25N3O2 |
| Exact Mass: | 315.19500 |
| Molecular Weight: | 315.41000 |
SMILES:
Saxagliptin, (1S,3S,5S)-2-(2S)-2-Amino-2-(3-hydroxyadamantan-1-yl)-acetyl)-2-azabicyclo[3.1.0]hexane-3-carbonitrile of the following chemical structure:
is a dipeptidyl peptidase IV (DPP4) inhibitor. Saxagliptin is marketed under the trade name ONGLYZA® by Bristol-Myers Squibb for the treatment of type 2 diabetes.
Saxagliptin and its hydrochloride and trifluoroacetic acid salts are disclosed in U.S. Pat. No. 6,395,767. In addition, U.S. Pat. No. 7,420,079 discloses Saxagliptin and its hydrochloride, trifluoroacetic acid and benzoate salts, as well as Saxagliptin monohydrate.
U.S. 2009/054303 and the corresponding WO 2008/131149 application disclose several crystalline forms of Saxagliptin and of Saxagliptin salts. The crystalline forms of Saxagliptin reported in that patent application are a monohydrate (denoted there as form H-1), a hemihydrate (denoted there as form H0.5-2), a dihydrate (denoted form H2-1) and an anhydrous form (denoted there as N-3).
WO 2005/117841 (the '841 application) describes the cyclization of Saxagliptin to form the therapeutically inactive cyclic amidine. The '841 application reports that such cyclization can occur both in solid state and solution state.
WO 2010/115974 discloses Forms: I-S, HT-S, IV-S, and HT-IV-S of Saxagliptin hydrochloride.

Org. Process Res. Dev., 2009, 13 (6), pp 1169–1176
DOI: 10.1021/op900226j

The commercial-scale synthesis of the DPP-IV inhibitor, saxagliptin (1), is described from the two unnatural amino acid derivatives 2 and 3. After the deprotection of 3, the core of 1 is formed by the amide coupling of amino acid 2 and methanoprolinamide 4. Subsequent dehydration of the primary amide and deprotection of the amine affords saxagliptin, 1. While acid salts of saxagliptin have proven to be stable in solution, synthesis of the desired free base monohydrate was challenging due to the thermodynamically favorable conversion of the free amine to the six-membered cyclic amidine 9. Significant process modifications were made late in development to enhance process robustness in preparation for the transition to commercial manufacturing. The impetus and rationale for those changes are explained herein.
Monohydrate 1 was isolated
as a white solid (58.2 kg, 88%).
1
H NMR (400 MHz, CD2Cl2-
d6) δ 5.25 (dd, J1 ) J2 ) 1.0 Hz, 1H), 4.93 (dd, J1 ) 10.6 Hz,
J2 ) 2.3 Hz, 1H), 3.55-3.50 (m, 1H), 3,35 (s, 1H), 2.45 (ddd,
J1 ) 16.1 Hz, J2 ) 10.9 Hz, J3 ) 5.6 Hz, 1H), 2.25 (dd, J1 )
13.6 Hz, J2 ) 2.5 Hz, 1H), 2.18-2.10 (m, 2H), 1.83-1.42 (m,
15H), 1.40-1.27 (m, 3H) 1.0-0.87 (m, 2H)
13C NMR (100
MHz, CD2Cl2) δ 173.43, 120.15, 68.83, 60.90, 46.57, 45.51,
45.08, 45.01, 41.62, 38.15, 37.92, 37.35, 35.88, 30.98, 30.93,
30.80, 18.00, 13.69.
MS (FAB) m/z 316 [M + H]+
1H NMR PREDICT


.................. http://www.google.com/patents/WO2012162507A1?cl=en
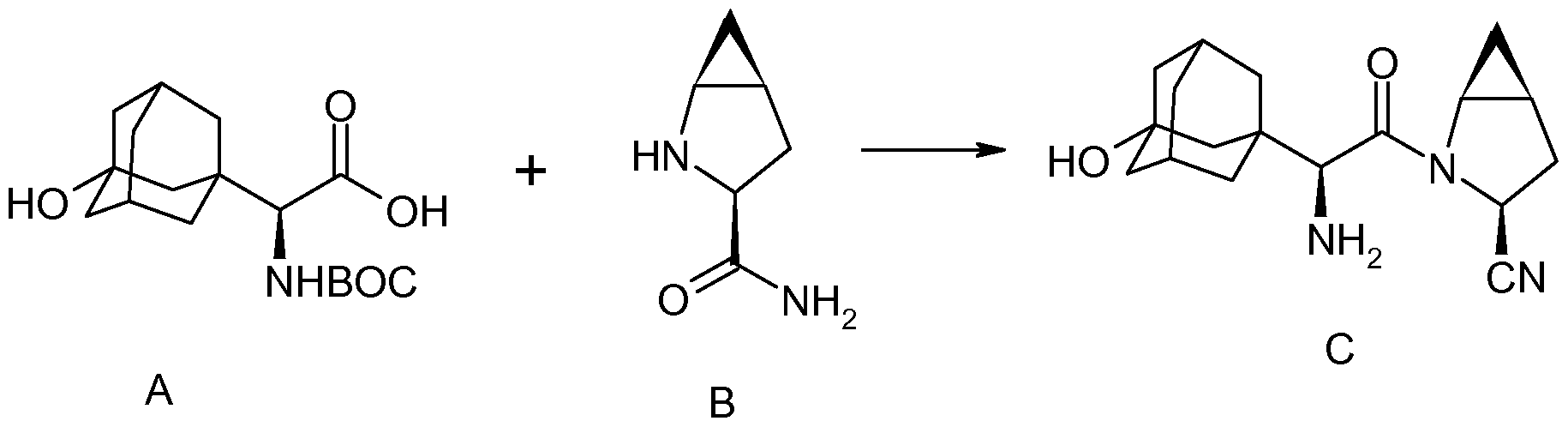
two amino acid derivatives (A) and (B), described in further detail hereinbelow, coupled in the presence of a coupling reagent. The amide coupling of (S)-a[[(l,l-dimethyleethoxy)carbonyl]amino]-3- hydroxytricyclo [3.3.1.1]decane-l-acetic acid (A) and (lS,3S,5S)-2-azabicyclo[3.1.0]hexane-3- carboxamide (B), subsequent dehydration of the primary amide and deprotection of the amine affords saxagliptin (C).
synthetic route is disclosed as follows:

Scheme-IV
.................. 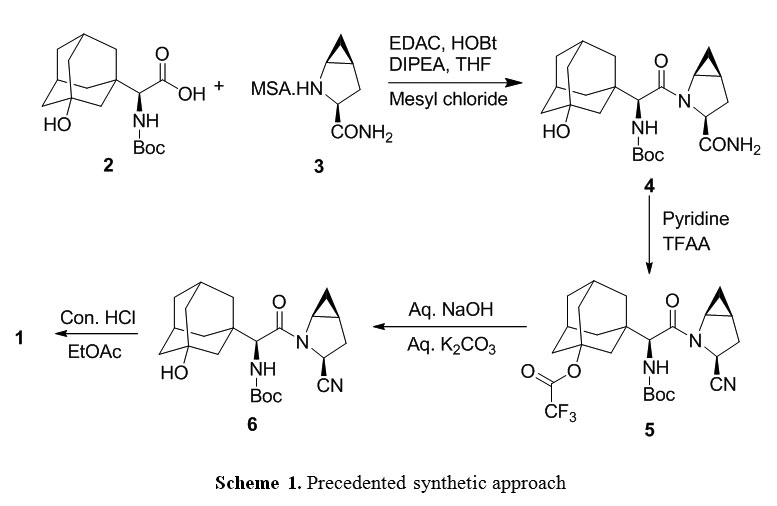 .................  .............   Savage, Scott A., et al., "Preparation of Saxagliptin, a Novel DPP-IV Inhibitor", Organic Process Research & Development, 2009, vol. 13, pp. 1169-1176. |
REFERENCES
| US6395767 | 16 Feb 2001 | 28 May 2002 | Bristol-Myers Squibb Company | Cyclopropyl-fused pyrrolidine-based inhibitors of dipeptidyl peptidase IV and method |
| US6995183 | 27 Jul 2004 | 7 Feb 2006 | Bristol Myers Squibb Company | Adamantylglycine-based inhibitors of dipeptidyl peptidase IV and methods |
| US7186846 | 28 Mar 2005 | 6 Mar 2007 | Bristol-Myers Squibb Company | Process for preparing a dipeptidyl peptidase IV inhibitor and intermediates employed therein |
| US7214702 | 23 May 2005 | 8 May 2007 | Bristol-Myers Squibb Company | Reacting the amide compound with phosphorus oxychloride in an organic solvent; treating the reaction mixture with water to form (1S,3S,5S)-2-[(2S)-2-amino-2-(3-hydroxytricyclo[3.3.1.13,7]dec-1-yl)-1-oxoethyl]-2-azabicyclo[3.1.0]hexane-3-carbonitrile-hydrochloride |
| US7223573 | 2 May 2005 | 29 May 2007 | Bristol-Myers Squibb Company | Enzymatic ammonolysis process for the preparation of intermediates for DPP IV inhibitors |
| US7420079 | 18 Nov 2003 | 2 Sep 2008 | Bristol-Myers Squibb Company | Intermediates for making 1(alpha-amino-1-(cyclopropyl-fused pyrrolidinylcarbonyl)methyl)-3-hydroxyadamantanes, e.g., methyl 3-hydroxy-<a-oxotricyclo[3.3.1.13,7]decane-1-acetate |
| US7470810 | 11 Jan 2005 | 30 Dec 2008 | Bristol-Myers Squibb Company | Such as 1-dodecane-thiotrifluoroacetate; alkyl/arylthiol is treated with trifluoroacetic anhydride in presence of pyridine, solvent (dichloromethane), and dimethylaminopyridine (DMAP) as catalyst; for protection of amino acids |
| US7741082 | 12 Apr 2005 | 22 Jun 2010 | Bristol-Myers Squibb Company | Process for preparing dipeptidyl peptidase IV inhibitors and intermediates therefor |
| US7943656 | 18 Apr 2008 | 17 May 2011 | Bristol-Myers Squibb Company | Crystal forms of saxagliptin and processes for preparing same |
| US20060035954 | 8 Aug 2005 | 16 Feb 2006 | Sharma Padam N | Ammonolysis process for the preparation of intermediates for DPP IV inhibitors |
| WO2001068603A2 | 5 Mar 2001 | 20 Sep 2001 | Bristol Myers Squibb Co | Cyclopropyl-fused pyrrolidine-based inhibitors of dipeptidyl iv, processes for their preparation, and their use |
| WO2008131149A2 | 18 Apr 2008 | 30 Oct 2008 | Squibb Bristol Myers Co | Crystal forms of saxagliptin and processes for preparing same |
| WO2010115974A1 | 9 Apr 2010 | 14 Oct 2010 | Sandoz Ag | Crystal forms of saxagliptin |
| WO2011140328A1 | 5 May 2011 | 10 Nov 2011 | Teva Pharmaceutical Industries Ltd. | Saxagliptin intermediates, saxagliptin polymorphs, and processes for preparation thereof |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US8748631 * | 24 May 2012 | 10 Jun 2014 | Apicore, Llc | Process for preparing saxagliptin and its novel intermediates useful in the synthesis thereof |
| US20130023671 * | 24 May 2012 | 24 Jan 2013 | Apicore, Llc | Process for preparing saxagliptin and its novel intermediates useful in the synthesis thereof |
- 1. Scott A. Savage, Gregory S. Jones, Sergei Kolotuchin, Shelly Ann Ramrattan, Truc Vu, and Rebert E. Waltermire (2009) Preparation of Saxagliptin, a Novel DPP-IV Inhibitor, Organic Process Research & Development., 13, 1169-1176.
- 2. Santosh K. Sing, Narendra Manne and Manojit Pal, (2008) Synthesis of (S)-1-(2-chloroacetyl)pyrrolidine-2-carbonitrile: A key intermediate for dipeptidyl peptidase IV inhibitors. Beilstein Journal of Organic Chemistry, 4, No. 20.
- 3. U.S. Pat. No. (2010) 0274025 A1.
- 4. U.S. Pat. No. (2006) 0035954 A1.
- 5. U.S. Pat. No. (2005) 0090539 A1.
- 6. Organic letters. (2001) Vol. 3, No.5, Page: 759-762
- 7. Tetrahedron 59 (2003) 2953-2989
 15
15 16
16 17
17
PART 1...........http://drugsynthesisint.blogspot.in/p/gliptin-series.html
PART 2 .......http://organicsynthesisinternational.blogspot.in/p/gliptin-series-22.html
SEE
PART 1...........http://drugsynthesisint.blogspot.in/p/gliptin-series.html
PART 2 ......http://organicsynthesisinternational.blogspot.in/p/gliptin-series-22.html
P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent.
//////////////////














No comments:
Post a Comment